Blogs & News
All FHIR’d up: Improving child health by optimising the use of clinical data
How can we improve the sharing of clinical data to benefit the patient directly and indirectly? That was the question posed to us last year when Aridhia were selected to be part of one of ten innovative healthcare data solutions in a UK-wide competition. The aim of this Sprint Exemplar was to develop proof of concepts for technology, methodology and research services that would inform the design of Health Data Research UK’s Digital Innovation Hub Programme.
The work was to take place at the newly established Digital Research, Informatics and Virtual Environments (DRIVE) unit at Great Ormond Street Children’s Hospital (GOSH). This unit was designed to use cutting-edge equipment to prototype and test new clinical technologies. It achieves this between the two teams there: the Clinical Informatics Research Programme (CIRP) and the team who manage the GOSH DRE (their specific deployment of our own Digital Research Environment, which is used to in research projects all across the Trust).
This blog focusses mainly on the FHIR implementation achieved at the DRIVE unit as part of this project. We approached the problem from two main angles:
- Make routinely collected clinical data available to researchers, securely and efficiently, while leveraging machine learning models.
- Make routinely collected clinical data and other patient data (e.g. from wearables and sensors) available to clinicians and patients via specialised applications on smartphones and tablets, using the Fast Healthcare Interoperability Resources (FHIR) and SMART on FHIR (SoF) standards.
First, what is SMART on FHIR?

FHIR is an increasingly widely used standard which describes data formats/elements, along with an application programming interface (API) for exchanging electronic health records.
Substitutable Medical Applications, Reusable Technologies (SMART) is an open standards-based platform used to securely develop apps on top of the FHIR API.
Here’s the important bit: Together, they create a framework to build healthcare applications that don’t rely on any specific vendor, so you get a seamless user experience for clinicians/patients, which is secure and runs across any healthcare system. You’re not having to write separate applications for each new system you encounter. This is helping us to achieve the “Interoperability” component of the FAIR Data Principles.
Project solution
As a research hospital, GOSH wants to allow clinicians and patients direct access to core clinical data, and as such, are very interested in developing innovative applications for clinical purposes. This data also includes data from wearables (such as Fitbits) and sensors, and their aim is to produce machine learning models which can be used to inform clinical care.
To meet this need we created the “SMART App Development Cycle”. Developers need to be able to develop SoF apps (which leverage the FHIR standard) within the controlled and secure Aridhia DRE Workspace environment.
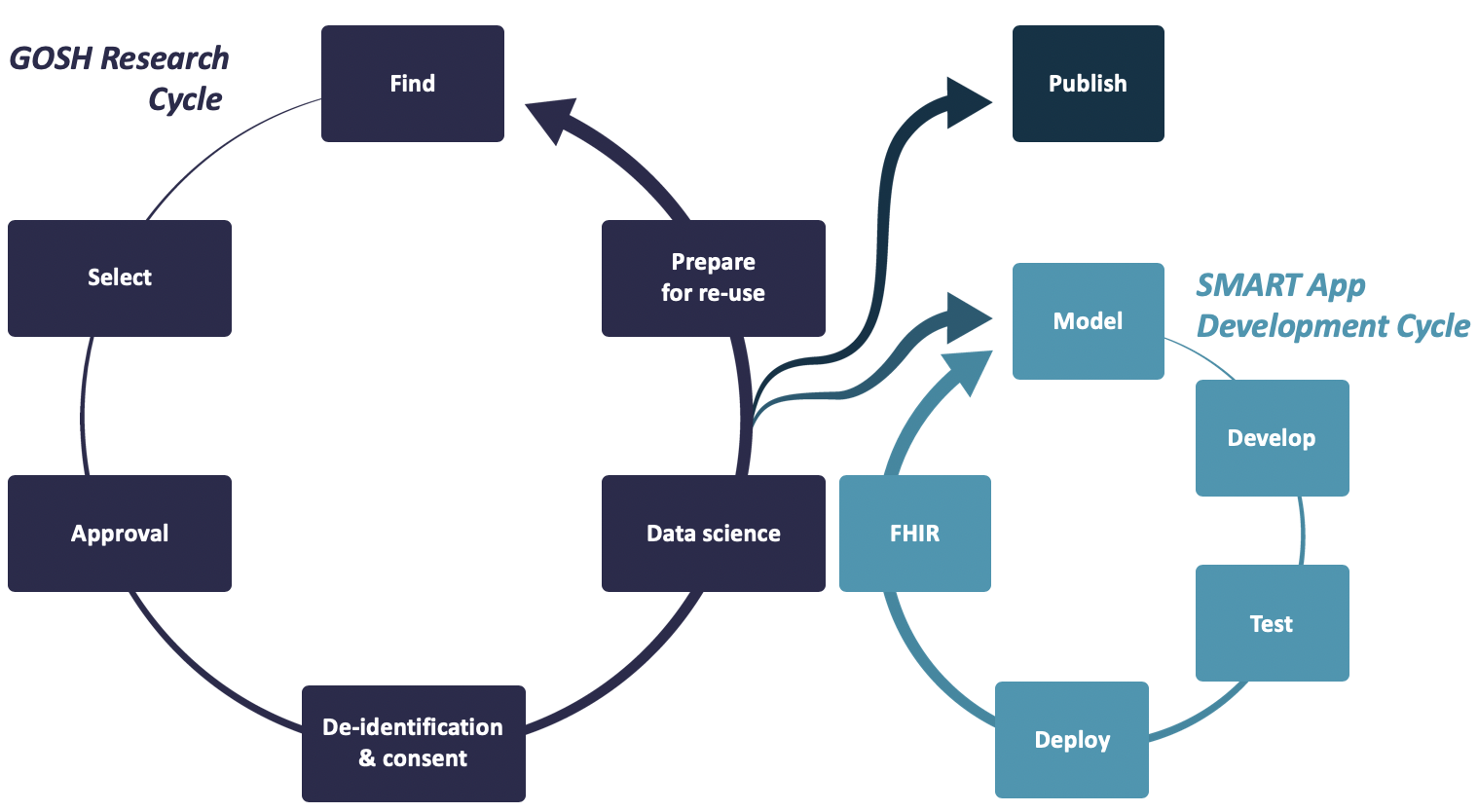
This prototyped process will now be the blueprint for future development within GOSH. Patients being able to view their health data on their smartphones will revolutionise the way GOSH and other research institutions deliver services. Using a global standard allows them to test innovations from partner hospitals around the world quickly and efficiently. Apps and machine learning models can be access and deployed via management layers on Microsoft Azure through the DRE.
Outcomes
By the end of the project we had managed to map and transform Cystic Fibrosis clinical data, along with Fitbit and sensor data into FHIR Objects, develop a FHIR Application for both patients and clinicians within a secure Workspace, and integrated a proof of concept machine learning model in the application. All this, plus also upgrading GOSH’s DRE FHIR service to the new FHIR API on Azure.
The end of project report, which goes into more detail, will be available soon. The full documented processes for SoF App development and machine learning to “applications in clinic” will soon be available as open-source on GitHub, so check back soon for a link to this.
If you’re interested in the Cystic Fibrosis project used as part of the test case here, you can find more information on Project Fizzyo here.
March 2, 2020
Alicia Gibson
Alicia Gibson joined Aridhia in 2018 and serves as the Chief Project Officer. With over 25 years of experience Alicia is a seasoned leader in project and operational management. With a wealth of experience in both internal and customer-facing projects she excels in aligning projects with the company’s vision. Alicia also leads the Customer Data Engineering team ensuring alignment with customer’s data engineering and data science needs and expectations. Prior to joining Aridhia Alicia worked at multiple Fortune 500 companies mainly in the technology sector and also joined an early-stage software startup. Alicia is a certified Project Manager and holds an MBA.
